A) 0.243 M
B) 0.200 M
C) 1.13 M
D) 0.563 M
E) 0.0313 M
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which element is reduced in the following reaction? Cu + 2H2SO4 → CuSO4 + SO2 + 2H2O
A) Cu
B) H
C) S
D) O
E) H2O
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is reduced in the following reaction? Cu(NO3) 2 + Zn → Zn(NO3) 2 + Cu
A) Cu(NO3) 2
B) N
C) O
D) Zn
E) NO3
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the net ionic equation if sodium sulfate is mixed with barium hydroxide?
A) 2Na+(aq) + SO42-(aq) → Na2SO4(s)
B) Na+(aq) + OH-(aq) → NaOH(s)
C) 2Ba+(aq) + SO42- (aq) → Ba2SO4(s)
D) Ba2+(aq) + SO42- (aq) → BaSO4(s)
E) Ba2+(aq) + 2OH-(aq) → Ba(OH) 2(s)
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a strong acid?
A) H3PO4
B) HNO3
C) HF
D) CH3COOH
E) H2O
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What mass of lithium phosphate is needed to prepare 500. mL of a solution having a lithium ion concentration of 0.125 M?
A) 2.41 g
B) 7.24 g
C) 14.5 g
D) 21.7 g
E) 43.4 g
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Suppose a large piece of a metallic solid, A(s) , is placed in a beaker containing the metallic cation Z2+(aq) , as depicted below. 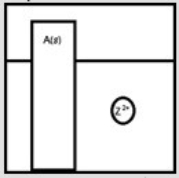 If Z is a more active metal than A, which is a possible representation of the final reaction mixture?
If Z is a more active metal than A, which is a possible representation of the final reaction mixture? 
A) I only
B) II only
C) III only
D) I or II
E) II or III
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Based on the solubility rules, which one of these compounds is soluble in water?
A) AgBr
B) AgCl
C) Ag2CO3
D) AgNO3
E) Ag2S
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
In each of the following cases, write down the oxidation number of the indicated atom. a. P in P4 b. C in C2H6 c. S in H2SO4 d. Mn in MnO4- e. P in Na3PO4
Correct Answer

verified
a = 0, b =...View Answer
Show Answer
Correct Answer
verified
View Answer
Short Answer
Identify the element being oxidized in the following reaction. 4Al + 3O2 → 2Al2O3
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the reaction depicted below, if Z represents Zn, which metal could A represent? 
A) Al
B) Na
C) Pb
D) Ca
E) Cu
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which chemical equation describes a combustion reaction?
A) 2C2H6(g) + 7O2(g) → 4CO2(g) + 6H2O(l)
B) LiOH(aq) + HNO3(aq) → LiNO3(aq) + H2O(l)
C) N2(g) + 3H2(g) → 2NH3(g)
D) 2Na(s) + 2H2O(l) �→ 2NaOH(aq) + H2(g)
E) 2Al(s) + 3H2SO4(aq) → Al2(SO4) 3(aq) + 3H2(g)
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is insoluble in water?
A) Li2CO3
B) NaOH
C) PbCl2
D) Ba(OH) 2
E) (NH4) 2S
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidizing agent in the following reaction? 5Fe2+(aq) + MnO4-(aq) + 8H+(aq) → 5Fe3+(aq) + Mn2+(aq) + 4H2O(l)
A) Fe2+
B) MnO4-
C) H+
D) Mn2+
E) Fe3+
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
35.0 mL of 0.255 M nitric acid is added to 45.0 mL of 0.328 M Mg(NO3) 2. What is the concentration of nitrate ion in the final solution?
A) 0.296 M
B) 0.481 M
C) 0.583 M
D) 0.911 M
E) 0.148 M
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Complete the following reaction and identify the Brønsted acid. NaOH(aq) + HCl(aq) →
A) NaH(aq) + HOCl(aq) ; NaOH is the acid.
B) NaH(aq) + HOCl(aq) ; HCl is the acid.
C) NaCl(aq) + H2O(l) ; NaOH is the acid.
D) NaCl(aq) + H2O(l) ; HCl is the acid.
E) NaCl(aq) + H2O(l) ; NaCl is the acid.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which represents the result of mixing equal volumes of 1 M aluminum chloride, 2 M magnesium chloride, and 1 M potassium chloride solution? (Each sphere represents 1 mol of ions.)
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which chemical equation describes an acid-base neutralization reaction?
A) 2Al(s) + 3H2SO4(aq) → Al2 (SO4) 3 (aq) + 3H2(g)
B) 2SO2(g) + 2H2O(l) + O2(g) → 2H2SO4(aq)
C) LiOH(aq) + HNO3(aq) → LiNO3(aq) + H2O(l)
D) 2KBr(aq) + Cl2(g) → 2KCl(aq) + Br2(aq)
E) CaBr2(aq) + H2SO4(aq) → CaSO4(s) + 2HBr(aq)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the pH of a 0.056 M HNO3 solution?
A) 0.056
B) 1.25
C) 12.75
D) 2.88
E) 11.11
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the oxidation number of N in K3Fe(CN) 6?
A) +3
B) -3
C) +4
D) -5
E) +1
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 146
Related Exams