A) Geometric
B) Optical
C) Linkage
D) Coordination
E) More than one of these
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the spectrochemical series, which one of the following ligands has the strongest field?
A) H 2O
B) CN −
C) NH 3
D) OH −
E) Cl −
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A characteristic of ligands is that
A) they are Lewis acids.
B) they are Lewis bases.
C) they are ions.
D) they are electron pair acceptors.
E) they are Brønsted-Lowry acids.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands could participate in linkage isomerism?
A) NH 3
B) H 2O
C) NH 4 +
D) NO 2 −
E) Ethylenediamine
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
Of the 3d transition series of elements, zinc has the greatest atomic radius.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The ground state electron configuration of a transition element atom cannot have more than one incomplete subshell.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The maximum oxidation state of an element in the first transition series never exceeds its group number.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most common oxidation state for ions of the transition elements is
A) +2.
B) +3.
C) +4.
D) +5.
E) +6.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following octahedral complex structures, each involving ethylene diamine and two different, unidentate ligands X and Y. 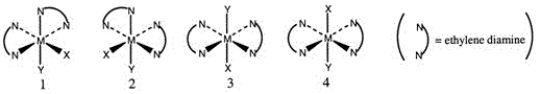 Which one, if any, of the following is a pair of optical isomers?
Which one, if any, of the following is a pair of optical isomers?
A) 1 and 2
B) 1 and 3
C) 1 and 4
D) 3 and 4
E) None of these choices are correct.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following complexes could have isomeric forms?
A) [Pt(NH 3) 3Cl] +
B) [Fe(CN) 6] 3 +
C) [Pt(NH 3) Br 3] +
D) [Pt(en) Cl 2]
E) [Co(H 2O) 4Cl 2] +
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands is most likely to form a low-spin octahedral complex with iron(III) ?
A) Cl −
B) H 2O
C) NH 3
D) OH −
E) CO
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following transition elements can have an oxidation number of +7?
A) V
B) Cr
C) Mn
D) Fe
E) Co
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the coordination number of cobalt in the complex ion [Co(en) Cl4]-? (en = ethylenediamine)
A) 1
B) 2
C) 4
D) 6
E) 8
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following coordination numbers applies to octahedral complexes?
A) 4
B) 5
C) 6
D) 8
E) None of these choices are correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
10.0 mL of a 0.100 mol/L solution of a metal ion M2+ is mixed with 10.0 mL of a 0.100 mol/L solution of a ligand L. A reaction occurs in which the product is ML3. Approximately, what is the maximum concentration of ML32+, in mol/L, which could result from this reaction?
A) 0.100
B) 0.050
C) 0.033
D) 0.025
E) 0.017
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following transition elements can achieve the largest oxidation number?
A) Chromium, Cr, Group 6B(6)
B) Manganese, Mn, Group 7B(7)
C) Iron, Fe, Group 8B(8)
D) Cobalt, Co, Group 8B(9)
E) Zinc, Zn, Group 2B(12)
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain transition metal complex has the formula MX42+. If the metal ion has a d8 electron configuration, what is the shape of the complex?
A) Octahedral
B) Square pyramid
C) Tetrahedral
D) Trigonal pyramid
E) Square planar
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions could exist in either the high-spin or low-spin state in an octahedral complex?
A) Sc 3+
B) Ni 2+
C) Mn 2+
D) Ti 4+
E) Zn 2+
G) A) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The Cu2+ ion has 1 unpaired electron.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands is most likely to form a high spin octahedral complex with cobalt(II) ?
A) CN −
B) en (ethylenediamine)
C) NO 2 −
D) CO
E) I −
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 81
Related Exams