A) square planar
B) tetrahedral
C) square pyramidal
D) see-saw
E) octahedral
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX4E will have a _____ molecular shape.
A) bent
B) see-saw
C) trigonal planar
D) T-shaped
E) square planar
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of N2O as predicted by the VSEPR theory? 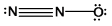
A) trigonal pyramidal
B) trigonal planar
C) angular
D) bent
E) linear
G) A) and B)
Correct Answer

verified
E
Correct Answer
verified
Multiple Choice
The formal charge on Cl in the structure shown for the perchlorate ion is: 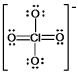
A) -2
B) -1
C) 0
D) +1
E) +2
G) None of the above
Correct Answer

verified
Correct Answer
verified
True/False
In formaldehyde, CH2O, both the formal charge and the oxidation number of carbon are zero.
B) False
Correct Answer

verified
Correct Answer
verified
Essay
A) Draw and name three molecular shapes for molecules having the VSEPR formulas AX3, AX3E and AX3E2, respectively. B) If the three X groups in the above formulas are identical, which of the three shapes would result in a molecule with a dipole moment?
Correct Answer

verified
a. The three structures and their molecular shapes are shown below. 11eb16b3_000b_7536_984d_270eb4710285_TB7799_00 b. The trigonal pyramidal and T-shaped molecules will have dipole moments.
Correct Answer
verified
Multiple Choice
What is the molecular shape of the thiocyanate anion, SCN¯, as predicted by the VSEPR theory? (Carbon is the central atom.)
A) linear
B) bent
C) angular
D) trigonal
E) None of these choices is correct.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the ideal bond angles around nitrogen in N2F2 using the molecular shape given by the VSEPR theory. (The two N atoms are the central atoms.)
A) 90
B) 109
C) 120
D) 180
E) between 120 and 180
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of NOCl as predicted by the VSEPR theory? 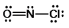
A) linear
B) trigonal planar
C) bent
D) tetrahedral
E) trigonal pyramidal
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
When resonance occurs, the bond lengths in a molecule fluctuate rapidly.
B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
In the nitrate ion (NO3¯) , nitrogen and oxygen are held together by
A) ionic interactions.
B) covalent bonds.
C) dative bonds.
D) electronegativity.
E) network bonds.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX3E2 will have a _____ molecular shape.
A) trigonal pyramidal
B) trigonal bipyramidal
C) trigonal planar
D) T-shaped
E) see-saw
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In which of the following does the nitrogen atom have a formal charge of -1?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to VSEPR theory, a molecule with the general formula AX5E will have a ______ molecular shape.
A) tetrahedral
B) trigonal bipyramidal
C) square pyramidal
D) octahedral
E) see-saw
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Essay
What is the shape of the PF3 molecule? Explain your answer, using VSEPR theory.
Correct Answer

verified
The Lewis structure has a lone pair on t...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
In which one of the following is the best Lewis structure a resonance structure?
A) CO2 (C = central atom)
B) ClO3¯ (Cl = central atom)
C) COCl2 (C = central atom)
D) NO2+ (N = central atom)
E) HCN (C = central atom)
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following Lewis structures is definitely incorrect?
A) NO ![]()
B) HCN ![]()
C) NO2¯ ![]()
D) SO32¯ ![]()
E) PCl5 ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the COCl2 molecule, carbon is the central atom. Based on the best Lewis structure for COCl2, what is the formal charge on carbon?
A) 0
B) +1
C) -1
D) +2
E) -2
G) D) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The Lewis structure of NO2 violates the octet rule.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the molecular shape of NO2¯ as predicted by the VSEPR theory?
A) linear
B) trigonal planar
C) bent
D) tetrahedral
E) resonant
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 108
Related Exams