A) +2.
B) +3.
C) +4.
D) +5.
E) +7.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A certain transition element has the stable oxidation states of +2, +3, +4, +5, and +6. In which state the element be most likely to form an ionic bond with chlorine?
A) +2
B) +3
C) +4
D) +5
E) +6
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the systematic name for [CoCl3(H2O) ]-.
A) cobalt(II) chloride monohydrate
B) aquatrichlorocobalt(II)
C) aquatrichlorocobaltate(II)
D) aquatrichlorocobaltite(I)
E) None of these choices are correct.
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The compound K3[Fe(CN) 6] is used in calico printing and wool dyeing. Give its systematic name.
A) potassium iron(III) hexacyanate
B) tripotassium iron(III) hexacyanate
C) potassium hexacyanoferrate(III)
D) potassium hexacyanideferrate
E) None of these choices are correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons will there be in a high-spin octahedral complex of Fe(II) ?
A) 0
B) 2
C) 4
D) 6
E) None of these choices are correct.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Consider the following structures (1 and 2 are octahedral; 3 and 4 are square planar) . 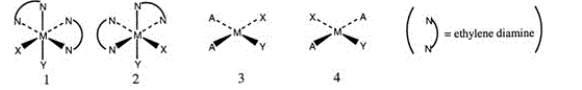 Which one of the following statements about the above structures is correct?
Which one of the following statements about the above structures is correct?
A) 1 and 2 are superimposable.
B) 1 and 2 are geometric isomers.
C) 3 and 4 are structural isomers.
D) 3 and 4 are optical isomers.
E) 3 and 4 are geometric isomers.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The most common oxidation state for ions of the transition elements is
A) +2.
B) +3.
C) +4.
D) +5.
E) +6.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following transition elements can achieve the largest oxidation number?
A) chromium, Cr, Group 6B(6)
B) manganese, Mn, Group 7B(7)
C) iron, Fe, Group 8B(8)
D) cobalt, Co, Group 8B(9)
E) zinc, Zn, Group 2B(12)
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be paramagnetic?
A) V5+
B) Ni2+
C) Mn7+
D) Ti4+
E) Zn
G) A) and E)
Correct Answer

verified
Correct Answer
verified
True/False
The conversion of the chromate ion (CrO42-) to the dichromate ion (Cr2O72-) is a redox process.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ligands is most likely to form a low-spin octahedral complex with iron(III) ?
A) Cl?
B) H2O
C) NH3
D) OH-
E) CO
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is considered a bidentate ligand?
A) cyanide, CN-
B) thiocyanate, SCN-
C) oxalate, C2O42-
D) nitrite, NO2-
E) hydroxide, OH-
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following will be the strongest oxidizing agent?
A) Cr
B) Cr(II)
C) Cr(III)
D) Cr(IV)
E) Cr(VI)
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the systematic name for Cr(CO) 3(NH3) 3.
A) chromiumtriaminotricarbonyl
B) triamminechromium carbonate
C) triamminetricarbonylchromate(0)
D) triamminetricarbonylchromium(0)
E) None of these choices are correct.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If M represents a transition element, which of the following oxides should be the least basic?
A) MO
B) M2O
C) M2O3
D) MO2
E) MO3
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The crystal field splitting energy, Δ,
A) is larger for tetrahedral complexes than for octahedral complexes.
B) depends on the metal but not on the ligand.
C) determines the color of a complex.
D) is larger for ionic ligands like chloride than for molecular ligands like carbon monoxide, CO.
E) determines the charge of a complex.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
According to valence bond theory, what would be the set of hybrid orbitals used when a Period 4 transition metal forms a tetrahedral complex?
A) d2sp
B) dsp2
C) dsp3
D) sp3
E) d2p2
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following ions could exist in only the high-spin state in an octahedral complex?
A) Cr2+
B) Mn4+
C) Fe3+
D) Co3+
E) Ni2+
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
The permanganate ion (MnO4-) is a powerful reducing agent.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following complexes could have isomeric forms?
A) [Pt(NH3) 3Cl]+
B) [Fe(CN) 6]3+
C) [Pt(NH3) Br3]+
D) [Pt(en) Cl2]
E) [Co(H2O) 4Cl2]+
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 40 of 82
Related Exams